Same Molecular Formula Different Structure
A molecular formula is a representation of a molecule that uses chemic symbols to indicate the types of atoms followed by subscripts to show the number of atoms of each type in the molecule. (A subscript is used merely when more than one atom of a given type is present.) Molecular formulas are also used equally abbreviations for the names of compounds.
The structural formula for a compound gives the same information as its molecular formula (the types and numbers of atoms in the molecule) but also shows how the atoms are continued in the molecule. The structural formula for methane contains symbols for one C atom and four H atoms, indicating the number of atoms in the molecule (Figure \(\PageIndex{i}\)). The lines correspond bonds that concur the atoms together. (A chemical bond is an attraction between atoms or ions that holds them together in a molecule or a crystal.) We will hash out chemical bonds and come across how to predict the organization of atoms in a molecule later. For now, simply know that the lines are an indication of how the atoms are connected in a molecule. A brawl-and-stick model shows the geometric arrangement of the atoms with diminutive sizes not to calibration, and a space-filling model shows the relative sizes of the atoms.
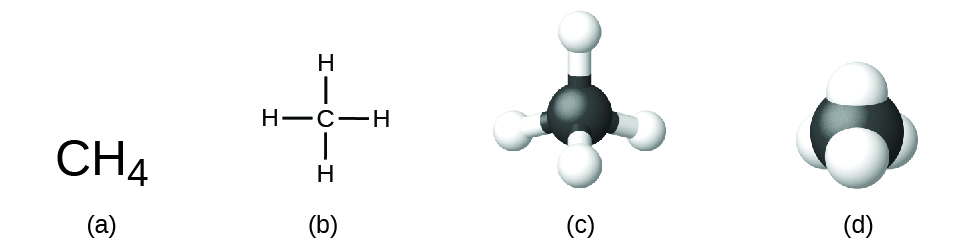
Figure \(\PageIndex{1}\): A methane molecule can exist represented as (a) a molecular formula, (b) a structural formula, (c) a ball-and-stick model, and (d) a space-filling model. Carbon and hydrogen atoms are represented by black and white spheres, respectively.
Although many elements consist of detached, individual atoms, some exist as molecules made up of two or more atoms of the element chemically bonded together. For instance, virtually samples of the elements hydrogen, oxygen, and nitrogen are composed of molecules that contain ii atoms each (called diatomic molecules) and thus have the molecular formulas H2, Oii, and N2, respectively. Other elements commonly found equally diatomic molecules are fluorine (F2), chlorine (Cl2), bromine (Br2), and iodine (I2). The most common form of the element sulfur is equanimous of molecules that consist of viii atoms of sulfur; its molecular formula is S8 (Figure \(\PageIndex{2}\)).
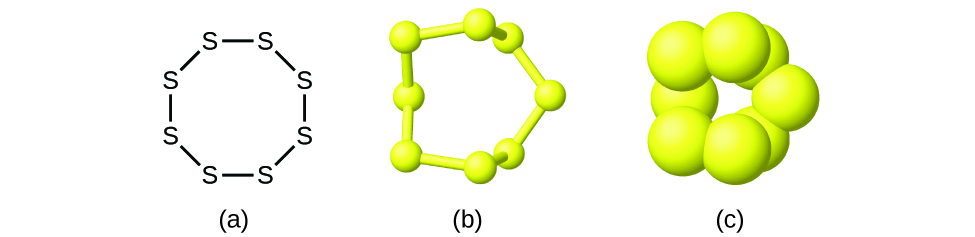
Figure \(\PageIndex{2}\): A molecule of sulfur is composed of eight sulfur atoms and is therefore written as S8. It tin be represented every bit (a) a structural formula, (b) a ball-and-stick model, and (c) a space-filling model. Sulfur atoms are represented by xanthous spheres.
It is important to notation that a subscript following a symbol and a number in front of a symbol practice not represent the aforementioned thing; for example, Htwo and 2H stand for distinctly dissimilar species. H2 is a molecular formula; it represents a diatomic molecule of hydrogen, consisting of two atoms of the chemical element that are chemically bonded together. The expression 2H, on the other hand, indicates 2 separate hydrogen atoms that are not combined as a unit. The expression 2H2 represents two molecules of diatomic hydrogen (Figure \(\PageIndex{three}\)).
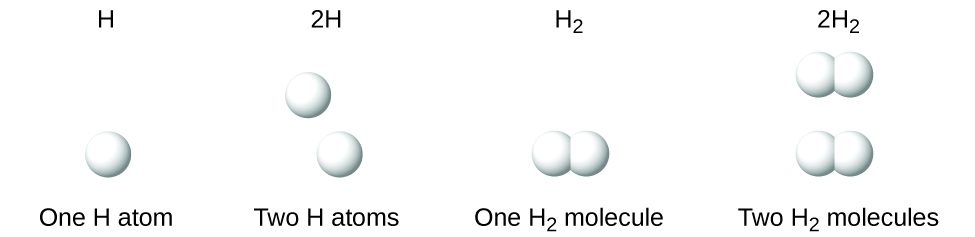
Effigy \(\PageIndex{3}\): The symbols H, 2H, Htwo, and 2Hii correspond very different entities.
Compounds are formed when two or more elements chemically combine, resulting in the formation of bonds. For example, hydrogen and oxygen can react to form water, and sodium and chlorine can react to form table common salt. We sometimes describe the composition of these compounds with an empirical formula, which indicates the types of atoms present and the simplest whole-number ratio of the number of atoms (or ions) in the compound. For example, titanium dioxide (used equally paint in white pigment and in the thick, white, blocking blazon of sunscreen) has an empirical formula of TiO2. This identifies the elements titanium (Ti) and oxygen (O) as the constituents of titanium dioxide, and indicates the presence of twice as many atoms of the element oxygen as atoms of the element titanium (Effigy \(\PageIndex{4}\)).
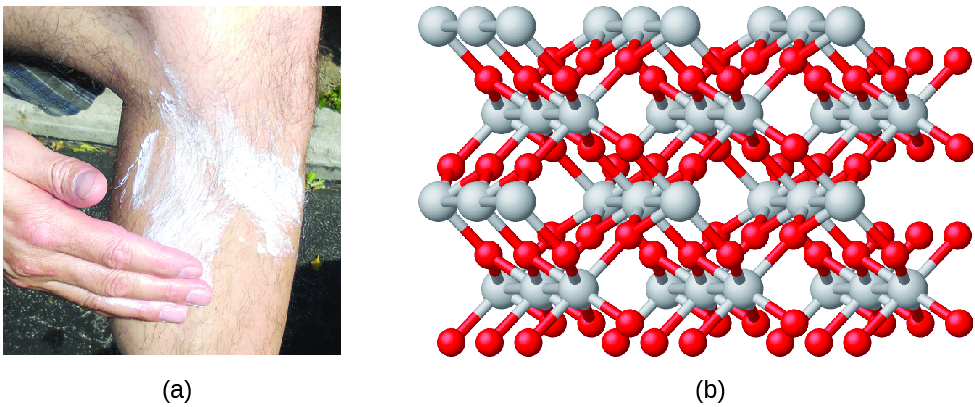
Figure \(\PageIndex{4}\): (a) The white chemical compound titanium dioxide provides effective protection from the sun. (b) A crystal of titanium dioxide, TiO2, contains titanium and oxygen in a ratio of ane to ii. The titanium atoms are gray and the oxygen atoms are cherry. (credit a: modification of work past "osseous"/Flickr).
Equally discussed previously, we tin can depict a chemical compound with a molecular formula, in which the subscripts indicate the bodily numbers of atoms of each element in a molecule of the compound. In many cases, the molecular formula of a substance is derived from experimental determination of both its empirical formula and its molecular mass (the sum of atomic masses for all atoms composing the molecule). For example, it can be determined experimentally that benzene contains ii elements, carbon (C) and hydrogen (H), and that for every carbon atom in benzene, there is one hydrogen atom. Thus, the empirical formula is CH. An experimental determination of the molecular mass reveals that a molecule of benzene contains six carbon atoms and six hydrogen atoms, and then the molecular formula for benzene is C6Hhalf-dozen (Effigy \(\PageIndex{5}\)).
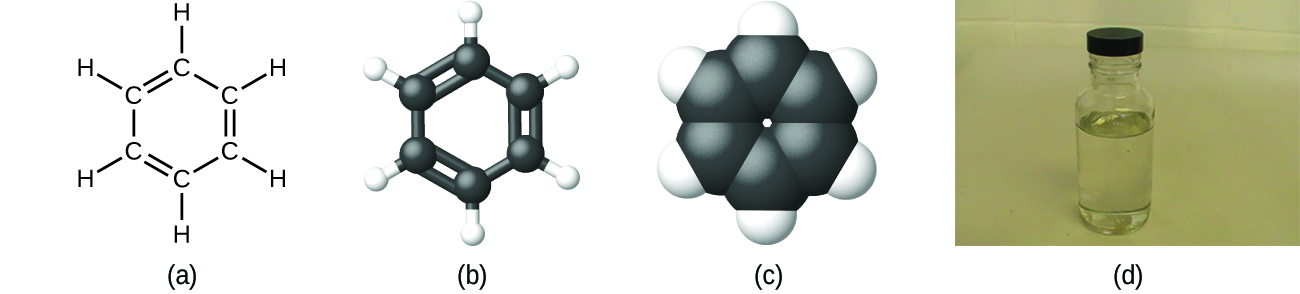
Figure \(\PageIndex{5}\): Benzene, C6Hhalf dozen, is produced during oil refining and has many industrial uses. A benzene molecule tin can be represented as (a) a structural formula, (b) a ball-and-stick model, and (c) a space-filling model. (d) Benzene is a clear liquid. (credit d: modification of work by Sahar Atwa).
If we know a chemical compound'southward formula, we can hands determine the empirical formula. (This is somewhat of an academic practice; the reverse chronology is generally followed in actual practice.) For case, the molecular formula for acerb acid, the component that gives vinegar its abrupt taste, is C2H4O2. This formula indicates that a molecule of acetic acrid (Figure \(\PageIndex{6}\)) contains ii carbon atoms, four hydrogen atoms, and 2 oxygen atoms. The ratio of atoms is 2:4:2. Dividing past the lowest mutual denominator (2) gives the simplest, whole-number ratio of atoms, 1:ii:one, and so the empirical formula is CHtwoO. Annotation that a molecular formula is always a whole-number multiple of an empirical formula.
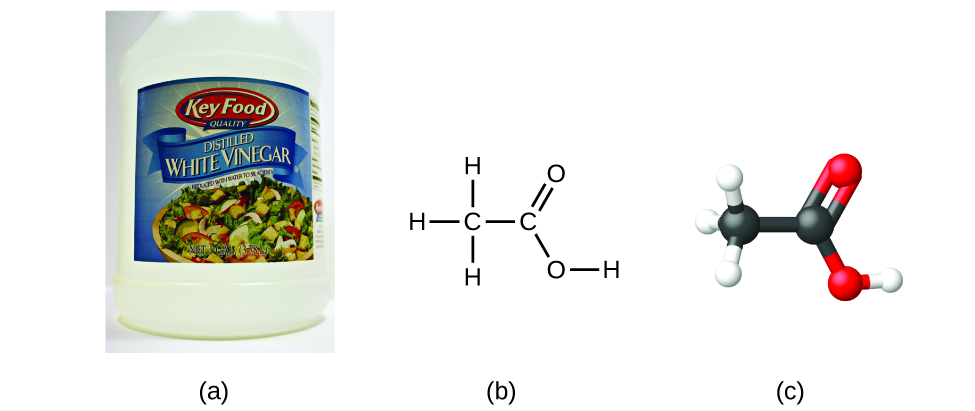
Figure \(\PageIndex{6}\): (a) Vinegar contains acetic acid, CtwoH4O2, which has an empirical formula of CH2O. It tin be represented every bit (b) a structural formula and (c) as a ball-and-stick model. (credit a: modification of piece of work by "HomeSpot HQ"/Flickr)
Example \(\PageIndex{ane}\): Empirical and Molecular Formulas
Molecules of glucose (blood sugar) comprise 6 carbon atoms, 12 hydrogen atoms, and 6 oxygen atoms. What are the molecular and empirical formulas of glucose?
Solution
The molecular formula is C6H12O6 because i molecule really contains 6 C, 12 H, and half-dozen O atoms. The simplest whole-number ratio of C to H to O atoms in glucose is 1:2:1, and then the empirical formula is CH2O.
Exercise \(\PageIndex{1}\)
A molecule of metaldehyde (a pesticide used for snails and slugs) contains 8 carbon atoms, 16 hydrogen atoms, and 4 oxygen atoms. What are the molecular and empirical formulas of metaldehyde?
- Answer
-
Molecular formula, CeightH16O4; empirical formula, CtwoH4O
Information technology is important to exist aware that it may be possible for the same atoms to be arranged in dissimilar ways: Compounds with the same molecular formula may have different atom-to-atom bonding and therefore different structures. For example, could there exist another compound with the same formula as acerb acid, C2H4Oii? And if so, what would be the construction of its molecules?
If yous predict that another compound with the formula CiiH4O2 could exist, then you demonstrated skilful chemical insight and are correct. Two C atoms, iv H atoms, and 2 O atoms can also be arranged to course methyl formate, which is used in manufacturing, as an insecticide, and for quick-drying finishes. Methyl formate molecules take 1 of the oxygen atoms between the two carbon atoms, differing from the arrangement in acetic acrid molecules. Acetic acrid and methyl formate are examples of isomers—compounds with the aforementioned chemical formula but unlike molecular structures (Figure \(\PageIndex{seven}\)). Note that this pocket-sized difference in the arrangement of the atoms has a major effect on their respective chemical properties. You would certainly not want to use a solution of methyl formate as a substitute for a solution of acetic acid (vinegar) when you make salad dressing.
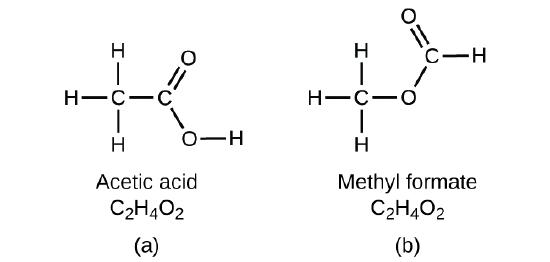
Figure \(\PageIndex{vii}\): Molecules of (a) acerb acid and methyl formate (b) are structural isomers; they accept the same formula (C2H4O2) but different structures (and therefore different chemical backdrop).
Many types of isomers be (Figure \(\PageIndex{8}\)). Acetic acid and methyl formate are structural isomers, compounds in which the molecules differ in how the atoms are connected to each other. There are also various types of spatial isomers, in which the relative orientations of the atoms in space tin be different. For example, the chemical compound carvone (found in caraway seeds, spearmint, and mandarin orange peels) consists of 2 isomers that are mirror images of each other. S-(+)-carvone smells like caraway, and R-(−)-carvone smells like spearmint.
Figure \(\PageIndex{8}\): Molecules of carvone are spatial isomers; they only differ in the relative orientations of the atoms in space. (credit bottom left: modification of piece of work by "Miansari66"/Wikimedia Commons; credit bottom right: modification of work by Forest & Kim Starr)
Summary
A molecular formula uses chemical symbols and subscripts to indicate the exact numbers of different atoms in a molecule or chemical compound. An empirical formula gives the simplest, whole-number ratio of atoms in a chemical compound. A structural formula indicates the bonding organisation of the atoms in the molecule. Ball-and-stick and space-filling models show the geometric system of atoms in a molecule. Isomers are compounds with the same molecular formula but different arrangements of atoms.
Glossary
- empirical formula
- formula showing the composition of a chemical compound given every bit the simplest whole-number ratio of atoms
- isomers
- compounds with the same chemical formula but different structures
- molecular formula
- formula indicating the composition of a molecule of a compound and giving the actual number of atoms of each element in a molecule of the compound.
- spatial isomers
- compounds in which the relative orientations of the atoms in space differ
- structural isomer
- 1 of two substances that have the same molecular formula but different physical and chemical properties considering their atoms are bonded differently
- structural formula
- shows the atoms in a molecule and how they are connected
Same Molecular Formula Different Structure,
Source: https://chem.libretexts.org/Courses/University_of_Kentucky/UK%3A_General_Chemistry/02%3A_Atoms_Molecules_and_Ions/2.4%3A_Chemical_Formulas
Posted by: hoagmusly1952.blogspot.com


0 Response to "Same Molecular Formula Different Structure"
Post a Comment